CREATING A NEXUS BETWEEN DISCOVERY AND COLLABORATION. Making the discoveries that advance human health is not simply a matter of bringing together the best minds from government, industry, academia and not-for-profits. It is also about knowing how to help those organizations collaborate in ways that harness their full power and potential. The FNIH excels at creating and then leading the processes that drive complex research projects forward. Whether we are working on a long-time health challenge or an emerging threat, we guide collaborations to achieve groundbreaking results that spur action, ultimately improving human health.
The Power of Collaboration:
Accelerating the Quest for Critical Data

Finding better markers of osteoarthritis
onset and progression
Osteoarthritis develops over time as a joint’s “cushion” of cartilage breaks down and bone ends rub together, causing pain and loss of function. Knee osteoarthritis (OA) is the leading cause of disability in older adults and affects about 250 million people worldwide. Right now, OA is diagnosed only after people have discomfort, and the standard tool is an X-ray showing joint space narrowing. But the disease process begins long before symptoms appear; identifying those at risk and intervening earlier could help avoid costly joint replacements.
Earlier diagnosis may soon be possible thanks to the Osteoarthritis Biomarkers Project, a $3.4 million study led by the FNIH Biomarkers Consortium in partnership with the National Institute of Arthritis and Musculoskeletal and Skin Diseases, the U.S. Food and Drug Administration (FDA), universities and pharmaceutical companies to identify biomarkers (biological markers) of knee OA onset and progression. Using data from 5,000 high-risk patients gathered through the National Institutes of Health Osteoarthritis Initiative, another public-private partnership that created an open-access repository of medical images and blood and urine samples, the FNIH-led team has narrowed thousands of potential biomarkers down to a subset that correlates with knee OA progression. These include bone and cartilage changes visible on MRI and proteins detectable in blood and urine. The biomarkers can pave the way toward identifying people at risk and developing treatments that target the disease process.
OSTEOARTHRITIS PARTNERS
AbbVie
Amgen
Arthritis Foundation
BIOIBERICA S.A.
DePuy Synthes Mitek Sports Medicine, a division of DePuy Orthopaedics, Inc.
U.S. Food and Drug Administration, Center for Devices and Radiologic Health
Flexion Therapeutics, Inc.
GlaxoSmithKline
Merck Serono
National Institute of Arthritis and Musculoskeletal and Skin Diseases
Rottapharm | Madaus
Sanofi
Identifying new targets to drive
drug development
Launched in 2014, the Accelerating Medicines Partnership (AMP) is a five-year, $230 million effort joining the resources of the FNIH, the National Institutes of Health (NIH) and the FDA with those of several nonprofit organizations and biopharmaceutical companies to devise a more precise approach to early-stage drug development. These organizations are sharing expertise and data to identify the biological pathways underlying four devastating diseases—type 2 diabetes (T2D), rheumatoid arthritis/systemic lupus erythmatosus (RA/SLE) and Alzheimer’s disease (AD)—and then validate the best candidates for targeting new medicines. AMP’s vision is to empower researchers to pursue new treatments with better odds of moving through the clinical trials pipeline successfully. Current therapies for these conditions cannot reverse the disease process nor stop its progression.
In 2015, the AMP-T2D Project publicly launched a knowledge portal that has amassed large amounts of genetic and clinical data from an array of studies on people with type 2 diabetes. The portal’s data cover a wide variety of ethnicities, including African-American, East Asian, South Asian, European and Hispanic, as well as diabetes-related cardiovascular and kidney complications. Researchers will be able to search and analyze the data to identify genetic variants associated with increased risk for, or protection against, the development of diabetes and its complications. In turn, these can become potential targets for new treatments.

The AMP-RA/SLE Project has developed new methods for acquiring and processing tissue from the kidneys and synovial membranes (linings of joints) and trained researchers to use these methods, making multi-site clinical studies possible. This tissue, along with blood and urine samples, is being utilized to generate a new public data repository to be launched in 2016. For the first time, partner organizations will be able to collaborate on genome-wide analysis of these samples to better understand the mechanisms behind the destruction caused by rheumatoid arthritis and systemic lupus erythmatosus, both diseases in which the body’s immune system destroys healthy tissue.
AMP-AD includes two different projects. Project A seeks to validate existing biomarkers as possible indicators of response to treatment in people with Alzheimer’s disease. Just one example is PET imaging of tau proteins in the brain, which has been added to two major NIH-funded phase II/III drug trials. Project B is working to accelerate the discovery of new therapeutic targets, moving beyond the traditional focus on the buildup of proteins such as amyloid and tau in brain tissue. Researchers build a deeper understanding of the networks in which these novel targets operate and validate them in different models. The project integrates analysis of large-scale molecular data from more than 2,000 human brains at all stages of the disease with network modeling approaches and experimental validation. This project has created a knowledge portal that enables rapid, broad sharing of data and analytical tools, and periodically releases public waves of large-scale molecular data from human brain samples. The ultimate goal is to shorten the time between discovery of potential drug targets to development of new drugs for Alzheimer’s treatment and prevention.
AMP PARTNERS
DIABETES:
American Diabetes Association
Eli Lilly and Company
Janssen Research & Development, LLC
JDRF International
Merck & Co., Inc.
Pfizer Inc
Sanofi
RA/SLE:
AbbVie
Alliance for Lupus Research
Arthritis Foundation
Bristol-Myers Squibb Company
The Lupus Foundation of America
Merck & Co., Inc.
Pfizer Inc
Rheumatology Research Foundation
Sanofi
Takeda Pharmaceuticals
ALZHEIMER’S DISEASE:
AbbVie
Alzheimer’s Association®
Alzheimer’s Drug Discovery Foundation
Biogen
Geoffrey Beene Foundation Alzheimer’s Initiative
GlaxoSmithKline
Janssen Research & Development, LLC
Eli Lilly and Company
Vradenburg Foundation
The Power of Innovative Models:
Reframing Approaches to Longstanding Challenges
Identifying biomarkers in children to help
advance autism research
An average of one in every 68 eight-year-old children in the United States is diagnosed with Autism Spectrum Disorder (ASD), typically based on behavioral observations by parents and caregivers—but these behaviors can range widely. Developing effective interventions and treatments through clinical trials depends on developing more sensitive and objective standards for diagnosing ASD in children and evaluating change over time.
In 2015, the FNIH partnered with the National Institute of Mental Health, Simons Foundation Autism Research Initiative (SFARI), FDA, Janssen Research & Development, LLC and European Autism Interventions – A Multicentre Study for Developing New Medications (EU-AIMS) to support a study of preschool and school-aged children with ASD to validate biomarkers that could help assess social function, track changes in behavior and neurologic function, and potentially evaluate the effectiveness of pharmacologic treatments. The FNIH is managing a $2 million contribution from SFARI to fund the Yale University study led by James McPartland, Ph.D., Principal Investigator, which is collecting data from children at five sites nationwide using assessments of social impairment, such as clinician, caregiver and lab-based tools; neurophysiological measures, such as eye tracking and studies of brain activity (EEG, or electroencephalogram); and blood samples from participants and their parents for genetic analysis. This $28 million large-scale U.S. partnership to identify biomarkers associated with ASD is part of the FNIH Biomarkers Consortium.
AUTISM PARTNERS
Eunice Kennedy Shriver National Institute of Child Health and Human Development
European Autism Interventions — A Multicentre Study for Developing New Medications (EU-AIMS)
Janssen Research & Development, LLC
National Institute of Mental Health
National Institute of Neurological Disorders and Stroke
Simons Foundation
Autism Research Initiative
U.S. Food and Drug Administration

Building research
infrastructure in Africa
To develop the new vaccines and treatments that improve public health, researchers require access to high-performance computing resources for collecting and analyzing complex biological data. Many low-income countries lack the infrastructure needed for bioinformatics research, even as they face some of the world’s most challenging and often-neglected diseases.
Last year, the FNIH played a pivotal role in helping the National Institute of Allergy and Infectious Diseases (NIAID) launch the first African Center of Excellence in Bioinformatics (ACE) in Mali, West Africa. Building on NIAID’s ongoing support for researchers in Mali, the FNIH invited corporate partners such as The BioTeam Inc., Intel Corporation Health and Life Sciences Group, the EMC Corporation and Hewlett Packard Enterprise to donate equipment for computing and data analysis, high-speed connectivity, and research and classroom spaces. Mali ACE opened in April 2015 at the University of Sciences, Techniques and Technology of Bamako (USTTB). Local researchers are now able to conduct advanced bioinformatics research while receiving long-distance training and mentorship by NIAID researchers. Mali ACE will serve as a model for future bioinformatics research centers in other regions of Africa.
ACE PARTNERS
The BioTeam Inc.
EMC Corporation
Hewlett Packard Enterprise
Intel Corporation Health and Life Sciences Group
National Institute of Allergy and Infectious Diseases
University of Sciences, Techniques, and Technology of Bamako, Mali, West Africa
Continuing in the search for
an HIV vaccine
Since the mid-1990s, the successful development of medications for people with human immunodeficiency virus, or HIV, has deflected some attention from the search for a preventive vaccine. But no medicine has cured HIV, and only about half of the more than 33 million HIV-positive people worldwide can access treatment. A successful vaccine is still considered fundamental for conquering HIV/AIDS.
For nearly 10 years, the FNIH has served as coordinator for the Comprehensive
T Cell Vaccine Immune Monitoring Consortium (CTVIMC), a central service facility within the Collaboration for AIDS Vaccine Discovery supported by the Bill & Melinda Gates Foundation. With more than $50 million in funding and researchers at multiple universities, as well as the Vaccine Research Center at the National Institute of Allergy and Infectious Diseases, the CTVIMC is looking at how candidate HIV vaccines trigger a response from the body’s immune system “defenders,” including T cells and B cells and the antibodies they activate. The FNIH’s role is to ensure that all of the researchers have access to a standardized set of T cell and B cell assays—including materials, techniques, measurements and reporting—so that results can be shared meaningfully across all sites. CTVIMC represents just one slice of the work going on within the Collaboration for AIDS Vaccine Discovery, which involves more than 600 investigators across 87 institutions in 12 countries.
CTVIMC PARTNERS
Becton, Dickinson and Company
Beth Israel Deaconess Medical Center
Biomedical Primate Research Centre
Case Western Reserve University
Centre hospitalier universitaire vaudois
Duke University
Fred Hutchinson Cancer Research Center
Henry M. Jackson Foundation
Imperial College London
International AIDS
Vaccine Initiative
McMaster University
National Institute of Allergy and Infectious Diseases
National Institute of Health and Medical Research (INSERM, France)
SeraCare Life Sciences
University of Montreal
University of Oxford
University of Washington
Vaccine & Gene Therapy Institute Florida
Wits Health Consortium
The Power of Support:
Channeling Resources for Greatest Impact

Recognizing groundbreaking work by
early-career scientists
Due to steady declines in financial support for basic science research, today’s young scientists face a challenging environment in which tenure-track university positions are scarce and about 85 percent of research grant proposals are unfunded. It is not unusual for biomedical researchers to reach their late 30s or 40s before receiving a major research grant—and this affects their ability to make significant, career-changing discoveries.
Three years ago, the FNIH established the Lurie Prize in Biomedical Sciences, named for philanthropist and Board member Ann Lurie, to help propel the work of one truly stellar, early-career researcher each year. The prestigious prize, which includes a medal and significant research honorarium, is awarded at a Washington, DC ceremony every spring. The 2015 Lurie Prize winner was Howard Hughes Medical Institute Investigator and Stanford University neuroscientist and psychiatrist Karl Deisseroth, M.D., Ph.D., who along with his team has pioneered two game-changing techniques: optogenetics, which uses light to map the neural circuits in our brains, and a technology called CLARITY that allows scientists to make organs transparent and therefore easier to study under a microscope. In his hands, both promise to revolutionize our approach to neurological diseases such as major depression, Alzheimer’s and Parkinson’s.
—(l to r) Solomon H. Snyder, M.D., Chairman, Lurie Prize Jury and FNIH Board Vice Chairman for Science; Ann Lurie, Lurie Prize Honorary Chair and FNIH Board Member; Karl Deisseroth, M.D., Ph.D., 2014 Lurie Prize Recipient; Maria C. Freire, Ph.D., FNIH President and Executive Director; Francis S. Collins, M.D., Ph.D., NIH Director and FNIH Board Ex Officio Member

Investigating the genetics of age-related eye disease
In 2001, researchers with the National Eye Institute’s Age-Related Eye Disease Study (AREDS) reported that a nutritional supplement called the “AREDS formulation”—including high doses of vitamin C, vitamin E, beta-carotene, zinc and copper—can reduce the risk of developing advanced age-related macular degeneration (AMD), the leading cause of vision loss and blindness for Americans over 65. A follow-up study called AREDS2 enrolled more than 4,200 people with AMD and found that replacing beta-carotene in the AREDS formulation with lutein and zeaxanthin provided an incremental beneficial effect and reduced the risk of lung cancer, especially in former and current smokers. Those results were published in 2013. Preventing the progression of AMD is critical since there are no effective treatment options for advanced AMD.
Thanks in part to support from the FNIH, the impact of the AREDS2 study will not end there. The FNIH provided funding to create the AREDS2 Genetic Repository, a collection of blood and saliva samples from AREDS2 participants. Genomic analysis is now under way, with the goal of understanding how a person’s genes play a role in the progression of vision-threatening AMD. Several genetic variants were already identified in a December 2015 Nature Genetics article. The repository is an important new tool for all researchers studying the genetics of AMD.
AREDS PARTNERS
Alcon Research Ltd.
Bausch & Lomb
Genentech
National Eye Institute

Preventing cognitive decline
Age-related decline in cognitive function is real and varies widely among individuals, influencing quality of life, character of personal relationships and capacity for making informed decisions about health care, retirement and other issues faced daily by millions of older adults.
In 2007, the McKnight Brain Research Foundation (MBRF) partnered with the FNIH and the National Institute on Aging (NIA) to fund the first Cognitive Aging Summit, where dozens of researchers shared ideas about how best to understand and remediate age-related cognitive decline. The Summit led to the Research Partnership in Cognitive Aging in 2008, a public-private effort between the NIA and the MBRF. Beginning in 2009, the partnership funded two research initiatives, 17 grants totaling $28 million over five years. The partnership supported a second Summit in 2010, with recommendations leading to a third joint initiative focused on interventions.
In 2014, the partnership, along with the National Center for Complementary & Integrative Health and the NIH Office of Behavioral and Social Sciences Research, funded the $15 million MEDEX (Mindfulness, Health Education, & Exercise) trial. Now underway, the trial is comparing the effectiveness of mindfulness-based stress reduction, brain health education and aerobic exercise as strategies for improving cognitive function.
COGNITIVE AGING PARTNERS
McKnight Brain Research Foundation
National Center for Complementary and Integrative Health
National Institute on Aging
NIH Office of Behavioral and Social Sciences Research
Supporting Patients and Education

EDMOND J. SAFRA FAMILY LODGE: TEN YEARS OF CARING
On November 5, 2015, the FNIH welcomed Board member and guest of honor Mrs. Lily Safra to celebrate the 10th anniversary of the Edmond J. Safra Family Lodge, which since 2005 has hosted more than 100,000 room nights for patients of the NIH Clinical Center and their families. The Family Lodge provides a home-like setting free of charge for guests from across the nation and around the world. This service exists thanks to the generosity of the Edmond J. Safra Philanthropic Foundation and many other individual donors, corporations and foundations. Mrs. Lily Safra donated proceeds from her Jewels for Hope auction to support
the preservation of the Family Lodge according to her vision.
— Mrs. Lily Safra (center) with the Family Lodge staff at the 10th Anniversary Open House Celebration

A GRATEFUL PATIENT’S PRAISE FOR RESEARCH
“As an engineer with a healthy dose of curiosity, the process of searching for answers has always interested me. I have been at the NIH on a daily basis for almost three months and have not only observed my caregivers taking copious notes based on what I’m experiencing, but also overheard post-doctoral interns talking to their advisors in the cafeteria, noticed the topics of numerous and daily seminars being displayed on the atrium monitors and developed a clear sense for everyone’s focus here. They are all searching for what they don’t know, challenging the norm and looking for answers in areas of human suffering that until even recently seemed completely mysterious, or at least decades away from any reliable solutions.”
“This constant drive, the never-ending curiosity to search and to search again and again—this is research! It is the bedrock of this amazing and unique institution. We need to make sure the curiosity and the drive to seek solutions never abate due to a lack of funding or worse, apathy.”
— Romit Bhattacharya of Morris Plains, New Jersey, who at the urging of family and friends, traveled 260 miles to the NIH during the summer of 2015 to receive treatment for stage III lung cancer as part of a clinical trial. The FNIH fondly remembers Mr. Bhattacharya, who passed away in March 2016

MEDICATION DONATIONS HELP REDIRECT FUNDS TO RESEARCH
Since 2009, Sanofi Foundation for North America and Novartis Pharmaceuticals Corporation have donated more than $6.4 million in medications to the NIH Clinical Center through the FNIH’s In-Kind Drug Donation Program, freeing up critical dollars for research. The center is the nation’s largest hospital entirely devoted to clinical research, and patients receive all care free of charge.

SUPPORT FOR A NEW GENERATION OF RESEARCH SCIENTISTS
In fall 2015, two young scholars at the NIH were awarded full funding for their first year of education and training, thanks to a generous gift to the FNIH by the Jayne Koskinas Ted Giovanis (JKTG) Foundation for Health and Policy.
Jose Delgado-Jimenez received an NIH Postbaccalaureate Intramural Research Training Award/Cancer Research Training Award (Postbac IRTA/CRTA), which offers recent college graduates who plan to apply to graduate or health professional school the opportunity to spend one or two years performing full-time research at the NIH. Jose has a special interest in nanotechnology and cancer therapeutics. The JKTG Foundation gift also supported a scholarship to NIH’s Graduate Partnership Program (GPP), which brings Ph.D. students to the NIH Intramural Research Program for dissertation research. Awardee Ryan Phillips’ research interests include mathematical/molecular modeling and brain circuitry.
Both students are working side-by-side with some of the leading scientists in the world and experiencing the collaborative, interdisciplinary research environment that NIH has to offer.
— (l to r) Ted Giovanis, FHFMA, MBA, President and Founder JKTG Foundation for Health and Policy; Jose Delgado-Jimenez; J. Graham Atkinson, D.Phil., JKTG Foundation, Exec. VP for Research and Policy; Martin Brechbiel, Ph.D., Section Chief Radiation Oncology Branch at the National Cancer Institute (Jose’s mentor)

MORE FELLOWSHIPS FOR KIDNEY CANCER RESEARCH
October 4, 2015, marked the 10th anniversary of the Boo! Run for Life event in Washington, DC to benefit the Dean R. O’Neill Renal Cell Cancer Fund. This fund, together with the Dr. Edward T. Rancic Memorial Cancer Research Fund, supports a rotation of full-time fellows in the laboratory of Dr. Richard Childs of the National Heart, Lung and Blood Institute, where they focus on developing novel therapies for advanced kidney cancer.
— Pictured: Jennifer Melis of
Washington, DC

NEW FUNDING, NEW CAREER PATHWAYS
Two major grants through the FNIH are helping the NIH create new career-building opportunities for aspiring researchers. Thanks to an $850,000 grant from the Amgen Foundation, the NIH is now one of a select group of higher learning institutions taking part in the Amgen Scholars Program, which provides hundreds of summer research opportunities to undergraduate students. A $200,000 gift from the Congress of Neurological Surgeons (CNS) has created the NINDS/CNS Getch Scholar Award in collaboration with the National Institute of Neurological Disorders and Stroke (NINDS). This competitive award will provide two years of funding to help an early-career neurosurgeon launch a clinical and research career at a U.S. academic medical center.
— 2015 NIH scholars of the Amgen
Scholars Program
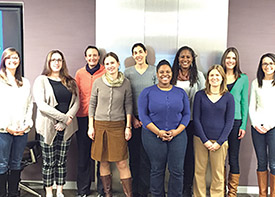
LAUNCHING WOMEN INTO RESEARCH CAREERS
In 2015, the Sallie Rosen Kaplan Postdoctoral Fellowship for Women Scientists in Cancer Research (SRK) Program welcomed 10 outstanding post-doctoral National Cancer Institute researchers—all female and some from underrepresented minority groups—to participate in leadership workshops and seminars, receive mentoring and coaching and develop a peer network. The program is designed to help with their transition to independent research careers.
—Pictured left to right: Elizabeth Spehalski, Rachel Van Duyne, Leah Randles, Mariia Novikova, Clara Bodelon, Khadijah Mitchell, Tiffany Lyle, Elizabeth Yanik, Pamela Gallagher, Maeve Mullooly

HUMAN GENOME EXHIBIT ON TOUR
Three million Smithsonian visitors toured Genome: Unlocking Life’s Code from June 2013 to August 2014 at the National Museum of Natural History, an exhibition made possible through funds raised, in part, by the FNIH. The result of a collaboration between the museum and the National Human Genome Research Institute, Genome showcased the complexity and power of the human genome using 3-D models, interactive displays, custom animations and real-life stories. The exhibit is now on a five-year tour to museums and science centers across North America.
—2015 HOST CITIES:
Portland, Saint Louis, San Diego
and San Jose
—2016 HOST CITIES:
Milwaukee (Jan. 23–April 25),
Salt Lake City (May 21–Sept. 5) and Wichita (Sept. 30 – Jan. 1, 2017)
